Sustainable Energy

Is green still greener?
$20+ million for cleaner, steady, and affordable ammonia supply
Everyone’s talking about ammonia. It’s a leading candidate in Earth’s race to decarbonize. The pungent gas does not emit carbon dioxide when burned, but making it requires electricity, which current manufacturers typically generate from fossil fuels like coal or natural gas. Prodromos Daoutidis is one of many researchers across the University of Minnesota who’s working on ways to green the process.
“Ammonia is really responsible for feeding the world,” said Daoutidis, professor of chemical engineering and materials science. “It’s produced at a massive scale—200 million tons per year—and it’s used as the basis for fertilizers that can dramatically increase crop yields. If you look at the rate that ammonia production is growing, it really follows the rate of the population growth.”
Yet, he adds, the industry is responsible for 500 metric tons of CO2 per year and nearly two percent of greenhouse gas emissions and fossil energy consumption.
Ammonia that’s 100 percent powered by renewables isn’t a new concept. It just hasn’t been a feasible, commercial option. Daoutidis hopes to change that.
“If you produce a chemical at a large scale, your rate of production is constant. It operates at a steady state as we say,” he explained. “In the case of green ammonia, because your feedstock is the wind or the sun, the feed stock fluctuates. This means you need a hugely oversized storage of hydrogen or a hugely oversized battery if you try to operate the system at a constant rate.”
His research group has developed new design and operation algorithms that adjust for intermittency.
“To build such a system, you cannot do a cookie-cutter design. You have to look at the particular location, wind or sun profile, and price of the electricity from that wind and sun capacity,” said Daoutidis, recipient of the 2023 AIChE Sustainable Engineering Forum Research Award for his significant technical contributions relative to the sustainability of products, processes, or the environment. “You also need to regularly adjust the level of production and create new safety precautions with regards to pressure or temperature, which will increase or decrease as you change the production rate.”
Demonstration studies will be conducted at the U’s West Central Research and Outreach Center inMorris—site of the world’s first wind-to-hydrogen and ammonia model plant.
The project builds on previous UMN green ammonia studies funded by the U.S. Department of Energy, totaling over $20 million. It also involves other colleagues in chemical engineering and materials (Qi Zhang, Alon McCormick, Paul Dauenhauer), plus faculty in mechanical engineering (Will Northrop), electrical engineering (Sairaj Dhople), and the Hubert H. Humphrey School of Public Affairs (E. Harrington).
International industry partners include Casale Switzerland and Shell.
“We definitely see potential for using renewable ammonia for large-scale storage of renewable energy, grid stabilization, regional-scale sustainable agriculture and energy, and community-scale applications,” Daoutidis said. “Minnesota has a huge tradition in ammonia. We have the experts and the infrastructure to be a world leader.”
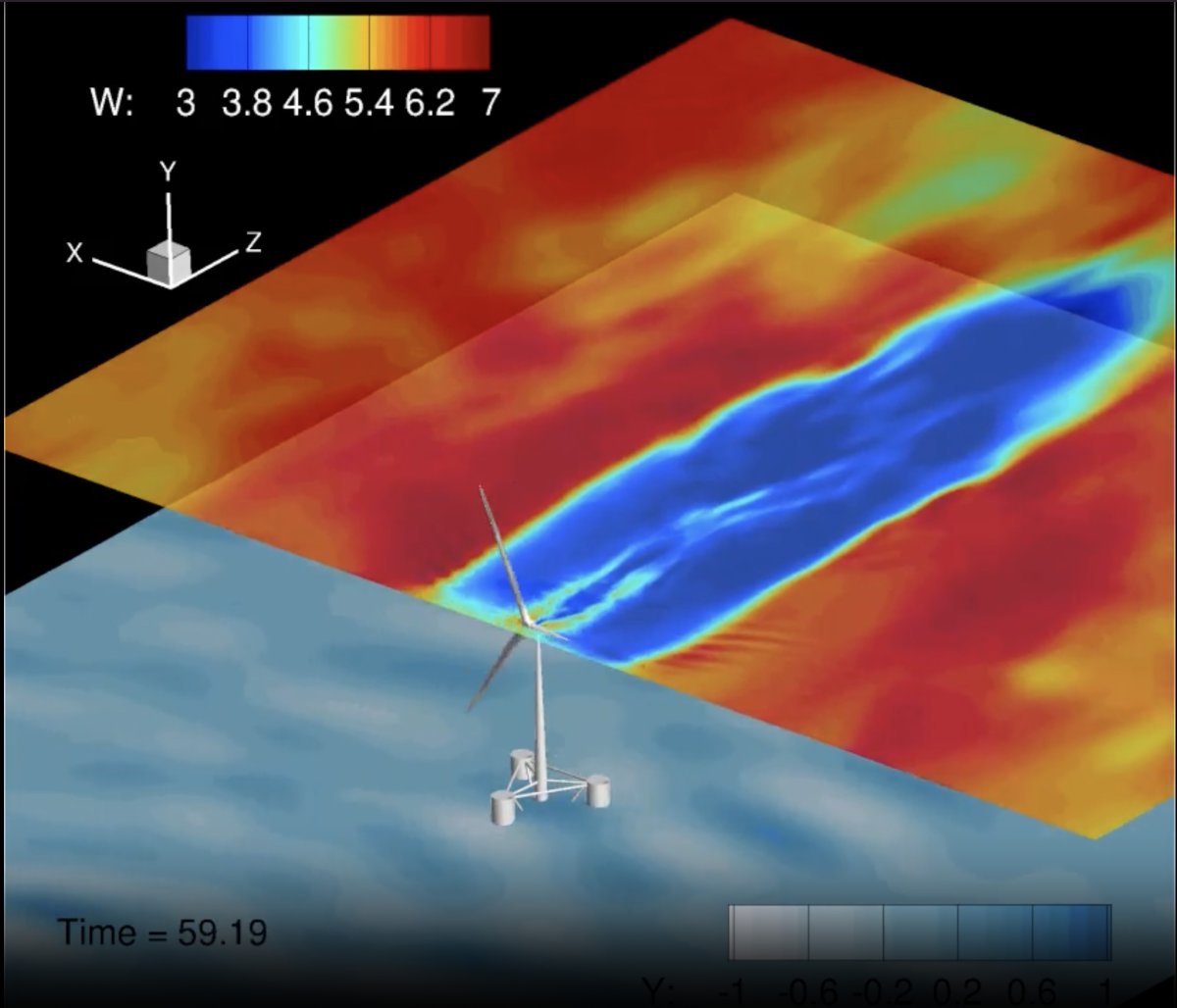
In deep, off shore
Simulations target wind turbine performance and stability
Nearly a decade ago, Lian Shen’s research group pioneered the use of computer simulation in the study of wind and wave interactions. Now he’s added another element to that mathematical modeling—a wind turbine that floats.
“Offshore wind energy is the next frontier. It is an important part in the future of renewable energy, with the potential to generate over 2,000 GW of capacity per year just in the U.S.,” said Shen, the Kenneth T. Whitby Professor of Mechanical Engineering who directs the U’s St. Anthony Falls Laboratory.
Most offshore turbines today are built in shallow waters or close to shore, with a fixed-bottom foundation and 6 to 10 megawatts (MW) of power. Shen and his colleagues are focused on those built in deep, open water. These 10 to 15 MW monsters with rotor diameters up to 240 meters will appear over the next 10 years, according to a report by the National Offshore Wind Research and Development Consortium, as shallow sites become scarcer.
“Before investing in these massive offshore wind turbine farms, it’s better to really understand them first. In this regard, a simulation is a very powerful research tool,” said Shen, who co-chairs the consortium’s R&D Advisory Group. “Our computer simulation is the first in the world that considers wind and waves, with the energy-carrying wind turbulence eddies and water wave components explicitly resolved, and their coupled nonlinear dynamics.”
Their study—backed by the National Science Foundation, U.S. Department of Energy, and New York State Energy Research and Development Authority—revealed the importance of ocean waves.
“We quantified the kind of impact, instantaneously and on average, they can impose on wind turbines,” Shen explained, “and how much energy can be harvested from an offshore wind farm.”
To a wind farm operator, this means an opportunity to minimize the impact of wake effects and potentially increase overall power output.
“The wind is in general stronger and steadier in the ocean than on land because it’s open—there are no mountains to block it. But offshore wind energy imposes a lot more challenges,” Shen said. “We wanted to simulate this on the computer, to sort of create this virtual reality first so, hopefully, we can provide some guidance for industry development in the coming years.”
Workforce for energy transition
CSE leads global community of scholars teaching electric power engineering
More than 400 faculty from 235- plus institutions across the United States have joined the Consortium of Universities for Sustainable Power (CUSPTM) to equip the next generation with the skills to steer the power grid and electricity usage toward a greener and more secure future.
Estimates suggest the demand for electric power engineers could be in the hundreds of thousands over the next 5 to 10 years, as the world combats climate crisis and industries jump on available clean energy incentives.
The 15-course CUSPTM online curriculum for workforce development was developed at the University of Minnesota Twin Cities and funded by federal agencies such as DOE, NSF, ONR, and NASA.
“The enrollments in power courses have dramatically declined nationwide, just when we need them evermore,” said Ned Mohan, a University of Minnesota electrical and computer engineering professor who leads the initiative. “By building this workforce we can provide a more sustainable future for our energy needs.”
Find out more: https://cusp.umn.edu
Patent Lesson
Researchers to trap CO2 with phosphate knowledge
Valérie Pierre knows a lot about phosphorous. The chemistry professor and her research team spent six years developing a process to detect it and remove it from surface water. Now she’s taking that patented knowledge and applying it to carbon dioxide (CO2).
Pierre has joined forces with two colleagues: Beth Stadler in electrical and computer engineering (also the college’s associate dean for academic affairs) and Emmanuel Detournay in civil, environmental, and geo- engineering (director of the U’s new $11 million Center for Interacting Geo-Processes in Mineral Carbon Storage).
“The challenge is how much do we capture and what do we do with it after we capture it,” Pierre said. “Fortunately, we’re not starting from zero. We’re starting from every success we’ve had with phosphate.”
That work—“Lanthanide Complexes for Catch-and-release of Phosphate Ions in Water”—is on its way to becoming a startup. Major companies are exploring partnering to commercialize it.
Pierre is also driven by the fact that “nobody’s done this,” she said. The general approach of others involves aggressive electro-chemicals and a lot of energy. “We’re proposing something different—low-energy, passive, scalable,” she added. “And we’re going for the oceans instead of lakes because oceans are bigger and there ’s 45 times more CO2 in them than air.”
Although only months into the research, the team has the molecules they need to experiment. “Now let’s see how much we can scale it up and optimize it,” Pierre said.
“That’s why we have this collaboration—Emmanuel is really good at doing this at a really gigantic scale; Beth has the expertise to make the structural materials for the chemical receptors that can catch and transport the carbon dioxide. Our team really highlights the strength of this college and what you can do when you have scientists working together with engineers.”